EU CLP Regulation (EC) No 1272/2008
Little Pro on 2015-12-30
The REGULATION (EC) No 1272/2008 on Classification, Labelling and Packaging of Substances and Mixtures, commonly known as CLP Regulation, entered into force on 20 January 2009. It aligns existing EU legislation to the United Nations Globally Harmonised System of Classification and Labelling of Chemicals (GHS).
The CLP Regulation also requires that suppliers of a hazardous substance (on its own or mixture) notify the European Chemicals Agency (ECHA) of its classification and labeling within one month of placing the substance on the market for the first time. This is often called C&L notification. Please note that there is no small volume exemption for C&L notification.
Critical Dates of CLP Implementation
| Substance |
|
| Mixture |
|
Classification
The classification of a substance or mixture reflects the type and severity of the hazards of that substance or mixture. Suppliers need to decide on the hazard class and hazard category of a substance or mixture by gathering available data and referring to chemical classification criteria given in CLP regulation. This is often called self-classification.
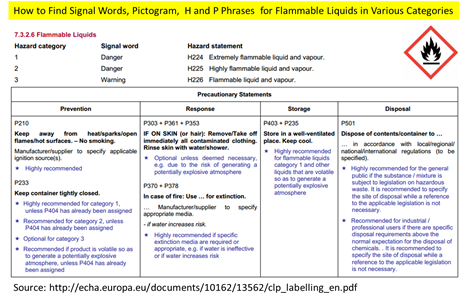
For example, a liquid with flash point between 23 and 60 Celsius degrees will be classified as Flammable Liquid Category 3. Based on this classification, you can easily find its signal word, pictogram, hazard statement, and precautionary statement (see picture below) in CLP regulation.
Currently CLP's classification criteria are aligned with UN GHS Rev. 5(8th ATP). Certain categories are not adopted such as acute toxicity category 5, flammable liquids category 4.
Annex VI to CLP - Compulsory Classification List
Annex VI of CLP includes harmonized classification and labelling for thousands of hazardous substances. It is mandatory for the suppliers of the respective substances or mixture to apply harmonized classification and labelling.
A few important notes when using compulsory classifications:
- For certain hazard classes, including acute toxicity and STOT repeated exposure, the classification in the Annex shall be considered as a minimum classification;
- For some entries (indicated by the reference ****), harmonized classification has not included physical hazards due to insufficient data. In this case, the correct classification for physical hazards shall be confirmed by testing;
- Please do read notes (i.e., A, B, P, H) for each entry which contain conditions for the harmonized classifications to apply;
Compulsory classifications in annex VI are available for from ECHA's classification &labelling inventory (C&L Inventory). C&L Inventory also contains classification and labelling information on notified and registered substances received from manufacturers and importers.
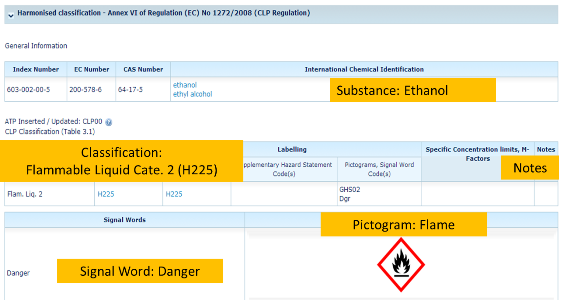
If you search ethanol (CAS 64-17-5), the following screenshot will pop up with harmonized classification for ethanol:
- Read more about annex VI to CLP regulation
- Factors to Consider When Using Harmonised Classifications in the CLP Annex VI
CLP Labelling Example
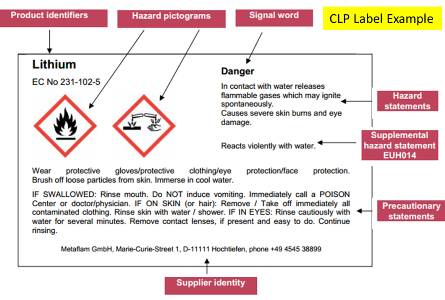
CLP label contains standard GHS label elements plus supplementary hazard info (please refer to special rules on packaging and labelling). The picture below is an example of a CLP label from ECHA's guidance:
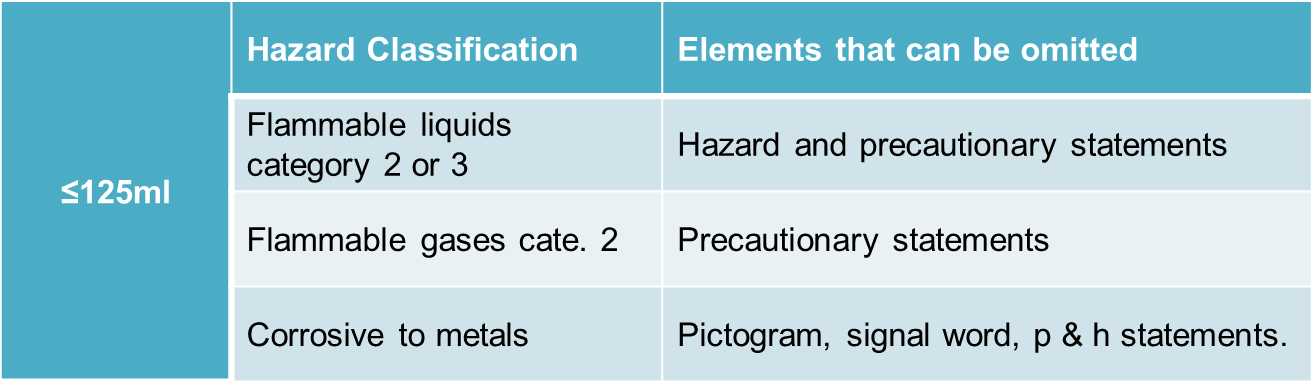
CLP regulation has also specified minimum dimensions of labels and pictograms under CLP for packages with different capacities (see picture below).
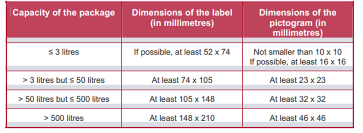
Other CLP labelling requirements include:
- 6 p-statements rule: Not more than six precautionary statements shall appear on the label unless it is necessary;
- Language: shall be prepared in the official language of destination Member States;
- Hiding composition info: A request for the use of an alternative chemical name shall be submitted to ECHA and approved by ECHA;
- Small packages (<=125mL): certain GHS label elements can be omitted (see example below);
Annex II to CLP - Special Rules on Packaging and Labelling
Supplemental hazard information which have not yet been included in the GHS but which have been part of the EU system in the past shall also be included in SDSs and on labels. A few examples are given below:
Example 1: Mixtures containing more than 0.15% lead by weight
EUH201- 'Contains lead. Should not be used on surfaces liable to be chewed or sucked by children' shall be labelled.
Example 2: Mixtures not classified as sensitising but containing at least one sensitising substance
EUH208-'Contains (name of sensitising substance). May produce an allergic reaction' shall be labelled.
Example 3: Labelling of plant protection products
EUH401-'To avoid risks to human health and the environment, comply with the instructions for use' shall be labelled.
In addition to that, CLP also requires child-resistant fastenings and tactile warning of danger on packages for certain hazardous chemicals.
| Child-resistant fastenings |
|
| Tactile danger warning |
|

C&L Notification
CLP regulation requires that information on substance identity, classification and labelling of a substance should be notified to ECHA within one month since a substance is placed on the EU market for the first time. C&L notification is very easy and it is usually done by manufactures or importers in the EU. There are various options to submit C&L notifications among which online submission may be the easiest. Please be noted that <1t/y non-hazardous substances do not need to be notified.
Safety Data Sheets
CLP classification and labelling info must be included in Safety Data Sheets. However, SDSs are governed by the REACH regulation. The Annex II of REACH has set out detailed requirements on the format and content of SDSs. Any new SDSs compiled or existing SDS updated after 1 June 2015 must conform to the requirements of the new version in Regulation (EU) 2015/830.
REACH SDS follows a 16 section GHS-format which is internationally agreed. The SDS must be supplied in an official language of the Member State(s) where the substance or mixture is placed on the market.
- Section 1: Registration number for substances subject to REACH registration must be provided; Email address mandatory;
- Section 8: List applicable Occupational Exposure Limits (OELs), Derived No-effect Level (DNEL) and PNEC;
- Section 15: Info on substances subject to authorization and restrictions; Indication of whether a chemical safety assessment has been conducted;
- eSDSs: Registrants and downstream users that are required to prepare a chemical safety report, must place the relevant exposure scenario(s) into an annex to the Safety Data Sheet.
Poison Centres notification (PCN)
If you place certain hazardous mixtures on the EU market (i.e. that are classified as hazardous on the basis of their health or physical effects), you will need to update your product labels and include unique formula identifier (UFI) on them. You will also need notify those hazardous mixtures (including the UFI) to the appointed bodies (i.e, poison centres) using the format and content harmonized by the amended Annex VIII of CLP regulation.
Read more: How to Comply with New Poison Centre Notification Requirements for Hazardous Mixtures in EU
Update of CLP Regulation via ATP
The technical annexes of CLP Regulation(for example, Annex VI), as well as some articles, need to be updated from time to time to ensure they are kept as comprehensive and accurate as possible. The amendments are known as adaptations to technical progress (ATPs) to the Regulation.
The most recent ATP is the 10th ATP amendment. The harmonised classifications within the 10th ATP will apply from 1 Dec 2018.
Reference & Resources
Click here to access all references and resources for Europe including the English translation of regulations, regulatory lists and useful links to the websites of competent authorities.
Recommended Reading
- Global GHS Implementation
- GHS label size requirements;
- GHS and confidential business info;
- GHS and pesticides;
- GHS classification list;
- GHS classification criteria in a single page;
- GHS regulations and standards;
- GHS label for small containers;
- Correlations between GHS and TDG
- Comparison of GHS building blocks in EU, USA, China and Japan
Having Questions?
We do not provide consultancy services. If you have questions or need any help, please contact our sponsor. You may also find an expert in CSP business directory below. If you are a consultant, you may get yourself listed in CSP business directory (free) or sponsor this page to leave your contact info on this page..

Tags: Topics - EU, GHS Implementation