How to Protect Confidential Business Info in Your SDSs?
Little Pro on 2016-02-16
GHS has given different competent authorities capabilities to make their own provisions to protect confidential business info in SDSs or on labels as along as such provisions do not compromise the health and the safety of workers and consumers. In some region or countries, withholding confidential business info in SDSs or labels even requires prior approval from local competent authorities. In this article, we have summarized the information disclosure requirements in major countries that have adopted GHS.
UN GHS
UN GHS purple book has set a few general principles for the disclosure of confidential business info (CBI) in SDSs and on labels:
- CBI claims should be limited to the names of substances and their concentrations in mixtures;
- Where CBI has been withheld, the SDS and the label shall indicate;
- CBI should be disclosed to competent authority upon request.
EU
In EU, essentially all hazardous substances must be declared in the safety data sheets (SDSs) if their concentrations exceed certain cut-off value (usually, 0.1% or 1% depending on hazards). Usually non-hazardous constituents do not need to be disclosed. However, non-hazardous substances must also be disclosed if there are union workplace exposure limits or if they belong to PBT substances. When disclosing composition info in SDSs, substance identifiers (CAS number or EC number), substance names as well as concentration or concentration ranges must be provided. If the concentration range is used, the classification derived for the particular concentration range should be based on the highest concentration in the range quoted.
Companies who are concerned about disclosing the full composition of a mixture in safety data sheets (SDSs) or on product labels can request the use of an alternative chemical name for a hazardous substance to protect their trade secrets. The request must be submitted to European Chemicals Agency (ECHA). A fee also needs to be paid in accordance with EU CLP regulation.
USA
According to OHSA’s hazard communication standards, the following info must be disclosed in SDSs and on labels:
- The chemical and common name(s) of all ingredients which have been determined to be health hazards, and which comprise 1% or greater of the composition, except that chemicals identified as carcinogens shall be listed if the concentrations are 0.1% or greater; and,
- The chemical and common name(s) of all ingredients which have been determined to be health hazards, and which comprise less than 1% (0.1% for carcinogens) of the mixture, if there is evidence that the ingredient(s) could be released from the mixture in concentrations which would exceed an established OSHA permissible exposure limit or ACGIH Threshold Limit Value, or could present a health risk to employees; and,
- The chemical and common name(s) of all ingredients which have been determined to present a physical hazard when present in the mixture;
Chemical name, substance identification number and exact weight percentage belong to trade secrets if the claim that the information withheld is a trade secret can be supported. Prior approval from OSHA is not required. All hazards must be disclosed. If confidential business info is claimed and withheld from SDSs, they must be indicated as trade secrets in SDSs.
Canada
According to Canada WHIMS 2015, for ingredients that present a health hazard, the chemical name, synonyms, CAS No. and exact concentration or specified concentration range must be given in SDSs [Ref]. Very broad concentration ranges are not allowed.
When a supplier or employer wants to be exempt from having to disclose confidential business information (CBI), such as the chemical identity of one or more trade-secret hazardous ingredients, they must file a claim for exemption with Health Canada. Once a complete application is received, a Hazardous Materials Information Review Act (HMIRA) Registry Number is assigned to the claim by Health Canada. The HMIRA Registry Number is required to be shown on the (material) safety data sheets ((M)SDSs) and for certain claims, on the label, for that product to be sold on the Canadian market or imported into Canada without disclosure of the CBI.
China
Both GB 15258-2009 and GB/T 17519-2013 have given clear guidance on information disclosure on labels and in SDSs. Hazardous ingredients contributing to the classification of a substance or mixture and their concentration or concentration ranges shall be disclosed. Generic names are acceptable.
For trade secret ingredients, real substance names and CAS numbers can be hidden. However, hazards shall be fully disclosed.
Japan
According to JIS7253, substance name and its concentration or concentration range shall be indicated in SDSs if it is present above concentration limit and contributes to the classification of a product.
If substance name belongs to trade secrets, a generic substance name can be used.However, the following substances must be disclosed and cannot be claimed as confidential:
- Respiratory sensitizing or skin sensitizing substance >0.1%w/w;
- Carcinogenic cate. 2 substance >0.1%w/w;
- Reproductive toxicant cate. 1 or cate. 2>0.1%w/w;
- STOT Cate. 2 substance >1%w/w;
Korea
The 2019 OSHA amendment requires companies to prepare safety data sheets for chemicals meet GHS hazard criteria, disclose full composition info and submit the safety data sheets to the MoEL prior to production or import. The SDS submission requirement will be enforced from 15 Jan 2021.
Hazardous substances must be fully disclosed in SDSs. If a company wishes to protect trade secret info and hide the real name of a hazardous ingredient, it must apply for prior approval from MoEL. If the application is granted, the company can then use a generic name instead of full name.
Taiwan
In Taiwan, hazardous ingredients contributing to the classification of a substance or mixture and their concentration shall be disclosed.
Companies who do not wish to disclose the name or concentration of hazardous chemicals or suppliers' names in SDSs for the necessity of national security or trade secret protection shall provide a written document to Taiwan OSHA and obtain an approval.
However, chemical substances with the following hazard classifications are not allowed to be withheld in safety data sheets (SDSs) and on labels:
- Acute toxicity cate. 1, 2 & 3;
- Skin corrosion and irritation cate. 1;
- Serious eye damage/irritation cate. 1;
- Respiratory or skin sensitisation;
- Carcinogenic, Mutagenic or Reproductive Toxicant;
- STOT single exposure or repeated exposure - cate. 1.
Australia
Australia requires that the chemical identity of a hazardous ingredient contributing to the classification of a chemical or a substance with occupational exposure limits be disclosed in safety data sheets (SDSs).
If substance name belongs to trade secret, the use of generic name for a hazardous substance is only accepted in certain cases (moderate hazard category and no occupational exposure limit established). If the exact concentration of an ingredient is confidential, the concentration of the ingredient can be disclosed using the following range or narrower range: <10%, 10-30%, 30%-60%, >60%.
Malaysia
In Malaysia, supplier may omit information on the name of a hazardous chemical or the composition and ingredients of a hazardous chemical in SDSs if the information belongs to confidential business information. If the substance name is omitted, such information shall be replaced with the generic names of the hazardous chemicals.
Where the exact concentration of an ingredient is CBI, the concentration of the ingredient shall be disclosed using the following allowable concentration range or a narrower range.
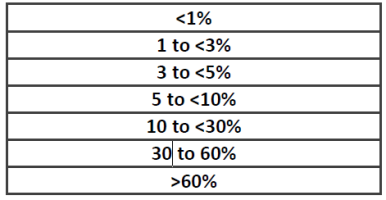
Where the classification of the hazardous chemical is based on the ingredients, rather than the product as a whole, the classification must be based on the highest concentration provided in the SDS.
Advanced GHS Readings
- GHS Precedence Rules for Pictogram, Signal Word and Hazard Statement
- GHS Classification of Mixture
- GHS Bridging Principles
- GHS Cut-off value/concentration Limit
- Global GHS Label Size Requirement
- Global GHS Label Requirement for Small Containers
- GHS and Pesticides
- GHS Classification List
- GHS and Confidential Business Info
- List of GHS Resources and Regulations
- Comparing GHS Builing Blocks in EU, USA, China and Japan
- Correlations between TDG and GHS
- When Should You Update Your SDSs
Having Questions?
We do not provide consultancy services. If you have questions or need any help, please contact our sponsor. You may also find an expert in CSP business directory below. If you are a consultant, you may get yourself listed in CSP business directory (free) or sponsor this page to leave your contact info on this page..

Tags: Topics - GHS, GHS SDS and Labelling