GHS Implementation in Canada
Little Pro on 2015-12-31
On 11 Feb 2015, Canada finally published its long-awaited Hazardous Products Regulations (HPR) which modified its Workplace Hazardous Materials Information System (WHMIS) to incorporate the Globally Harmonized System of Classification and Labelling of Chemicals (GHS) for workplace chemicals.
Manufacturers or importers will be required to classify and label hazardous chemicals in accordance with the new GHS-based hazard communication system (called WHMIS 2015) from 1 June 2017 (postponed to 1 July 2018 due to industry's challenges). In this article, we have analyzed chemical classification, labelling and SDS requirements in WHIMS 2015 and compared it with OSHA's Hazard Communication Standards (HCS 2012) in the United States.
Transitional Periods of WHMIS 2015/Canada GHS
GHS will be implemented in Canada in three phases. Different deadlines apply to manufacturers/importers, distributors and employers. The picture below summarizes the transitional period for the implementation of WHMIS 2015.
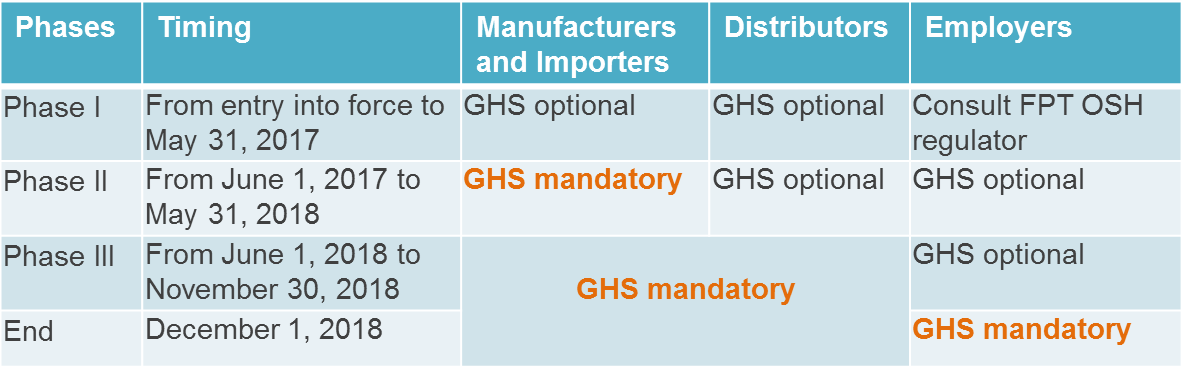
Chemical Classification Criteria in WHMIS 2015/Canada GHS
- Has not adopted environmental hazards and acute toxicity category 5; [Ref]
- Non-GHS hazard classes introduced for:
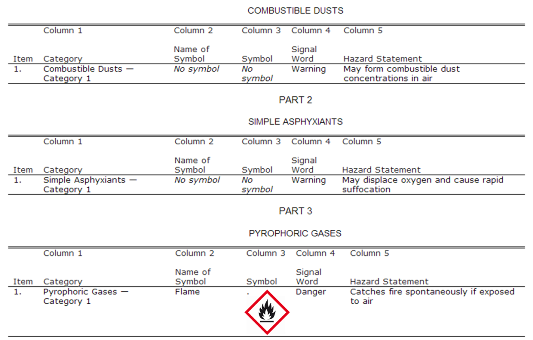
- COMBUSTIBLE DUSTS - please refer to the picture below;
- SIMPLE ASPHYXIANTS;
- PYROPHORIC GASES;
- PHYSICAL HAZARDS NOT OTHERWISE CLASSIFIED - no standard hazard statements;
- BIOHAZARDOUS INFECTIOUS MATERIALS - a new pictogram introduced;
- HEALTH HAZARDS NOT OTHERWISE CLASSIFIED;
Labelling Requirements in WHMIS 2015/Canada GHS
- Standard GHS labelling elements: hatched border no longer required; [Ref]
- Product Identifier: disclosure of hazardous ingredients in a mixture not mandatory;
- Pictogram: environmental pictogram is not mandatory;
- Additional pictogram required for "Biohazardous Infectious Materials" if applicable (please see the picture below);
- Hazard statement: additional statements for non-GHS hazard classes;
- Precautionary statement: backslash or diagonal mark [/] or three full stops [...] in a precautionary statement must not appear;
- Supplier info: must be the contact info of a Canadian manufacturer or importer;
- Language: must be in both English and French;
- Supplemental statements required for:
- a hazardous product which releases a gaseous substance that meets the classification criteria for Acute Toxicity in contact with water; or
- a mixture classified in the Acute Toxicity hazard class and containing ingredients of unknown acute toxicity.

Special Rules on Labelling in Canada WHMIS 2015
- Small capacity containers (100 ml or less): precautionary or hazard statements not required;
- Small capacity containers (3 ml or less): labels not required during their use stage;
- Bulk shipment and unpackaged hazardous products: labels not required;
- In transit products or export-only: WHIMS labels and SDSs are not required;
- Other special rules for lab samples and complex mixtures. [Ref]
Safety Data Sheets in WHMIS 2015
- Material Safety Data Sheets (MSDS) will be renamed as Safety Data Sheets (SDSs);
- Standard 16-section GHS SDSs;
- Section 2: must also include non-GHS hazard classes if applicable;
- Section 3: In the case of a hazardous mixture, each ingredient that, individually, presents a health hazard must be listed;
- SDSs must be in both English and French - a bilingual or two separate ones;
- Confidential info: use of concentration range accepted [ref]. However, if suppliers wish to withhold the chemical identity of hazardous ingredients, they must file a CBI exemption and pay a fee.
Canada WHMIS 2015 vs HCS 2012
While most of SDSs and labels that are compliant with the U.S. Hazard Communication Standard (2012) can be used in Canada, there are still some differences that suppliers need to know when complying with WHMIS 2015. Luckily, Health Canada has summarized the variances so that we do not need to find the differences by ourselves. Please click the picture below to download the comparison between WHMIS 2015 and HCS 2012.
Related Regulations and Standards
- Canda WHMIS Full Regulation
- OSHA's Hazard Communication Standards (2012);
- Hazardous Materials Identification System (HMIS) ;
- NFPA Ratings and NFPA 704 Labels;
- Globally Harmonized System of Classification and Labelling of Chemicals (GHS);
Recommended Reading
Having Questions?
We do not provide consultancy services. If you have questions or need any help, please contact our sponsor. You may also find an expert in CSP business directory below. If you are a consultant, you may get yourself listed in CSP business directory (free) or sponsor this page to leave your contact info on this page..

Tags: Topics - Canada, GHS Implementation
