Chemical Risk Assessment: Overview and Examples
Little Pro on 2016-05-17
In this article, we will give you an introduction to chemical risk assessment. We will focus on EU REACH regulation and industrial chemicals. However, the principles given in this article are consistent with risk assessment methodologies for other sector uses such as pesticides and cosmetics.
If you have no chemistry, toxiclogy or eco-toxicology background, please start with following parts first.
- Chemical risk assessment basics part 1: Physico-chemical properties
- Chemical risk assessment basics part 2: Ecotoxicology and e-fate studies
- Chemical risk assessment basics part 3: Toxicology studies
Risk = Hazard x Exposure
When it comes to chemical management, a distinction must be made between hazard and risk. Hazard defines the inherent property of a chemical having the potential to cause adverse effects when an organism, system or population is exposed to that agent. Risk however, establishes the probability of the adverse effect occurring.
To be more specific, the risk of a chemical depends on the following 2 factors:
- The inherent toxicity of the chemical (hazard);
- How much of a chemical is present in an environmental medium (e.g., water, soil, air) and how much contact a person or ecological receptor has with the chemical substance (exposure).
A hazardous chemical substance poses no risk if there is no exposure. For example, sulfuric acid is very corrosive. It is of no or little risk to ordinary people who do not handle them. For some people who may be exposed to sulfuric acid (scientists, workers), risk management measures (i.e, wearing goggles and gloves) can be taken to minimize the risk.
Chemical Risk Assessment: Purpose, Procedure and Tasks
The goal of chemical risk assessment is to have a full understanding of the nature, magnitude and probability of a potential adverse health or environmental effect of a chemical. It takes into account of both hazard and exposure. Risk assessment forms the foundation of regulatory decisions for industrial chemicals, pesticides, pharmaceuticals, cosmetics, food additives and food contact substances in developed countries today.
In general, chemical risk assessment consists of the following three steps:
- Hazard characterization: Dose-response determination (LD50/LC50, NOAEL, T25, EC50, NOEC, etc), determining the relationship between the magnitude of exposure to a hazard and the probability and severity of adverse effects.
- Exposure assessment: identifying the extent to which exposure actually occurs. Exposure levels are usually estimated or measured.
- Risk characterization: combining the information from the hazard characterization and the exposure assessment in order to form a conclusion about the nature and magnitude of risk, and, if indicated, implement additional risk management measures.
The picture below summarizes the complete procedure of chemical risk assessment under REACH.

The table below summarizes the detailed tasks of hazard characterization, exposure assessment and risk characterization for human health and the environment.
| Tasks | Human Health | Environment |
|---|---|---|
| Hazard Characterization |
Derive GHS classification for both acute/chronic effects, and local/systemic effects. Evaluate how mobile the substance is (e.g. volatility, water solubility, dustiness) and, depending on the uses, which routes of exposure; Derive no-effect levels for human health (DNEL) from available toxicology studies; take into account the foreseeable routes of exposure and populations. When no DNEL can be derived a qualitative or semi-quantitative characterization should be made. |
Derive GHS classification; Determine whether the substance should be a PBT substance; Evaluate in which environmental compartment the substance will predominantly end up (e.g. degradability and distribution behavior); Derive Predicted-No-Effect-Concentration (PNEC) for various environmental compartments from available eco-toxicity studies. |
| Exposure Assessment |
Gather info on operational conditions (for example, the duration and frequency of use or the amount used) and risk management measures (e.g. local exhaust ventilation or a certain type of glove). Calculate exposure estimate for potential routes of exposure and populations • Workers: dermal and inhalation. • Consumers: oral, dermal and inhalation |
Gather info on operational conditions (for example, the amount produced/used and emission percentage) and risk management measures (e.g. waste treatment measures). Estimate Predicted Environmental Concentration (PEC) for potential environmental compartments or use measured data. • Fresh surface water • Marine surface water • Soil • Sediment • STP micro-organism • Air • Predator |
| Risk Characterization |
Risk characterization ratio (RCR) = Exposure Estimate/DNEL • RCR<1, acceptable risk; • RCR>1, unacceptable risk. |
Risk characterization ratio (RCR) = PEC/PNEC • RCR<1, acceptable risk; • RCR>1, unacceptable risk. |
It shall be noted that risk characterization is an iterative process. If initial RCR>1, certain risk management measures can be taken to effectively reduce the RCR by reducing exposure. Such measures may include reducing the amount used, reducing the time or frequency of working, or reducing emissions.
How to Obtain Derived No- Effect Level (DNEL)?
Derived No- Effect Level (DNEL) is the level of exposure to the substance above which humans should not be exposed. The DNELs are calculated by dividing the value of the health effect dose descriptor (NOAEL, NOAEC, LD50, LC50) by an assessment factor .Since dose descriptors are obtained from experimental data, an assessment factor is required to allow for extrapolation to real human exposure situations.
It may not always be possible to derive DNELs for each health effect. This may be the case, for example, for carcinogenicity, where no safe threshold level can be obtained. In these cases a semi-quantitative value, known as the DMEL or Derived Minimal Effect level may be developed
Read More: How to Derive DNELs? How to Calculate DMELs?
DNEL and Human Healh Risk Assessment Example
| Effects | Adrenal Effects) | Developmental Effects |
|---|---|---|
| NOAEL from animal study (oral route ) | 30 mg/kg bw/day (90d repeated dose) | 80 mg/kg bw/day |
| Assessment Factor (Intraspecies) | 10 | 10 |
| Assessment Factor (Interspecies) | 10 | 10 |
| Assessment Factor (Duration) | 3 | 1 |
| Assessment Factor (Route extrapolation, data quality) | 1 | 2 |
| Total Assessment Factor (AF) | 10x10x3x1=300 | 10x10x1x2=200 |
| DNEL(oral route, long-term) | 0.1mg/kg bw/day | 0.4 mg/kg bw/day |
In above case, the DNEL used for risk characterization will be 0.1mg/kg bw/day. If an adult (assuming weight is 60kgs) intakes 12mg of a chemical substance per day, the estimated exposure (external dose per body weight) will be 0.2mg/kg bw/day. Since the exposure estimate is greater than DNEL, which will lead to a RCR>1, the risk will not be acceptable.
Note: Assessment factors are not randomly chosen. For more info about how to choose appropriate assessment factors to obtain DNEL, please read how to derive DNEL.
How to Obtain Predicted No Effect Concentration (PNEC)?
The Predicted No Effect Concentration or PNEC is the concentration of a substance in any environment below which adverse effects will most likely not occur during long term or short term exposure. The PNEC needs to be determined for each environmental compartments (water, soil, sediment, etc.).
The PNEC for each environment is estimated by dividing the dose descriptor by the relevant assessment factor. Since dose descriptors are obtained from laboratory tests involving a limited number of species, the assessment factor is required to account for the uncertainties involved in the extrapolation to the real ecosystems.
Where several dose descriptors are available for an environment, all the possible PNECs will be derived.The lowest PNEC will later be used for risk characterization.
PNEC and Environmental Risk Assessment Example
| Compartment | Eco-toxicology Dose Descriptors | Assessment Factor | PNEC value |
|---|---|---|---|
| Surface water |
|
10 | 1mg/L |
| STP-microorganism | 3h-NOEC>1000mg/L (activated sludge inhibition test) | 10 | 100mg/L |
| Soil | LC50 (earthworm acute toxicity) >1000mg/kg | 1000 | 1mg/kg |
In above case, PNEC-surface water is calculated as 1mg/L based on test data on the most sensitive species (Daphnia). Let's assume that we discharge waste water containing 20mg/L of a substance directly to river and the dilution factor is 10, the Predicted Environmental Concentration (PEC-water) will be 2mg/L. Since the RCR(equal to PEC-water/PNEC-water) is 2, the risk is not acceptable. However, if we take some risk management measures (i.e, oxidization, neutralization) to remove >60% of the substance from waste water before discharging, we will get a RCR less than 1 and acceptable risks.
For other environmental compartments (sediment, soil, STP), we can do the same risk characterization. For more info about how to choose appropriate assessment factors to derive PNECs for various environmental compartments , please read how to derive Predicted No Effect Concentration (PNEC).
How to Estimate Exposure Level or PEC?
Under REACH, exposure estimation is only required for hazardous substances. When estimating exposure, all human populations liable to exposure and all environmental compartments for which exposure to the substance is known, need to be addressed.
Ideally, the process for estimating exposure should be based on measurement data. In practice, the availability of reliable measurement exposure data is scarce and mostly limited to the workplace. In most cases, exposure estimation has to be based on exposure estimation models.
The most convenient models (also free) include:
- ECETOC TRA model for workers and consumer exposure estimation
- EUSES model for environmental exposure estimation
The reason that ECETOC TRA and EUSES are widely used is because they use standard parameters in a typical exposure scenario as input data. The standard parameters include the use descriptors, the concentration of a substance in a product, the applied amount, the duration of exposure or the presence of local exhaust ventilation or personal protection equipment.
If you could gather all standard parameters for each exposure scenarios/use listed in the table below and input them to ECETOC TRA and EUSES, you will get exposure estimates as output data.
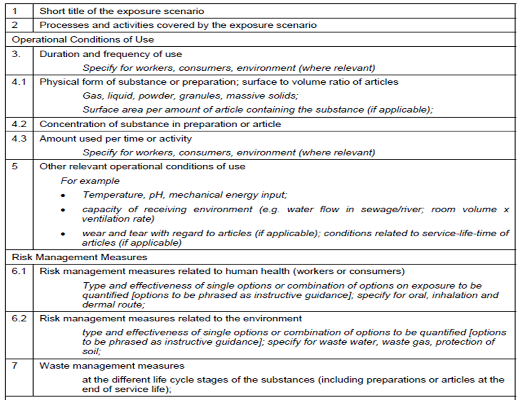
It should be noted that each of these models offers an initial estimation of exposure based on conservative or worst case exposure conditions. This estimation is usually defined as Tier 1 estimation. When the risk characterization shows that risks are not under control for these exposure conditions, additional estimation based on more detailed and specific data may be needed. This higher Tier estimation can be done using more sophisticated and detailed models or measured exposure data.
For more info about how to estimate exposure levels, please read the articles below.
- How to use ECETOC TRA to estimate worker exposure and consumer exposure (being developed);
- How to use EUSES to estimate environmental exposure
References and More Readings
- ECHA Guidance on Chemical Safety Assessment (n a Nutshell)
- ECHA Guidance on Information Requirements and Chemical Safety Assessment
- ICCA Guidance on Chemical Risk Assessment

Good job. You have learned the difference between hazard and risk. You have also learned what are tasks of hazard characterization, exposure assessment and risk characterization. Please keep it going. This is part 4 of our chemical risk assessment basics.
"It does not matter how slowly you go as long as you do not stop. "
– Confucius
Having Questions?
We do not provide consultancy services. If you have questions or need any help, please contact our sponsor. You may also find an expert in CSP business directory below. If you are a consultant, you may get yourself listed in CSP business directory (free) or sponsor this page to leave your contact info on this page..

Tags: Topics - CRA, Chemical Risk Assessment Tutorials