What Is Benchmark Dose (BMD) and How to Calculate BMDL
Little Pro on 2018-08-01
In chemical risk assessment, both no-observed-adverse-effect level (NOAEL) and statistical benchmark dose (BMD) can be used as point of departure (POD) to derive human health guidance value such as reference dose (RfD) or derived no-effect level (DNEL) or acceptable daily intake (ADI). Nowadays the BMD approach has become the US EPA’s preferred dose–response assessment method. Other authorities such as European Food Safety Authority (EFSA) also use the BMD method for food safety risk assessment. In this article, we will give you an introduction to the benchmark dose (BMD) approach and show to how to calculate benchmark dose (lower confidence limit), also known as BMDL.
What Is Benchmark Dose (BMD) and Benchmark Response (BMR)
A benchmark dose (BMD) is a dose or concentration that produces a predetermined change in the response rate of an adverse effect. This predetermined change in response is called the benchmark response (BMR).
Normally, the default BMR is 5% or 10% change in the response rate of an adverse effect relative to the response of control group depending on whether response data is continouous or quantal(dichotomous).
| Response Data | Examples | Default BMR |
|---|---|---|
|
Continuous data The values of a biological parameter in individual animals |
|
5% (EFSA) 10% (EPA) |
|
Quantal data The fraction of animals with a specific effect in a population |
|
10% |
After a critical effect and the BMR have been determined, different mathematical models are applied to fit the dose-reponse data and estimate the benchmark dose (BMD).
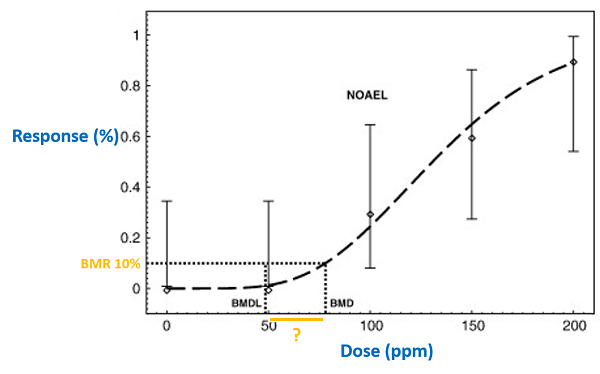
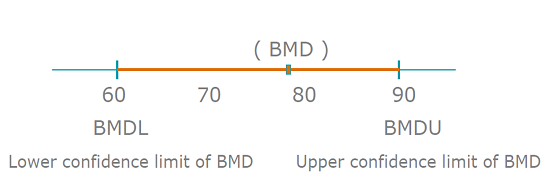
It should be noted that each fitted (and accepted) model results in a confidence interval for the estimated BMD. The BMD we get from statistical model is a range, rather than a fixed number (please see picture below). Usually we use benchmark dose (lower confidence limit) (BMDL) to calculate human health guidance value since it is more conservative. The BMDL can be regarded as a dose at which the effect is smaller than the BMR (with defined confidence).
How to Use BMDL in Risk Assessment
The main goal of calculating BMDL is to use it to estimate a daily oral or dermal exposure level to the human population (including sensitive subgroups) that is likely to be without an appreciable risk of deleterious effects during a lifetime.
- RfD or DNEL or ADI = POD (NOAEL or BMDL)/UF(Uncertainty factors)
Note: UF ranges from 100 to 10,00. Read more..
After RfD or DNEL has been derived, it will be compared with acutual or estimated human exposure level to determine if exposure risk is acceptable or not.
Comparison of BMD Method and NOAEL Approach
BMDL is more reliable than NOAEL since it is less dependent on dose selection and sample size. J. Allen Davis, Jeffrey S.Gift and Q. Jay Zhao from the US EPA have made a very good comparison of BMD method and traditional NOAEL approach in their research paper [Read..]
| BMD advantages | NOAEL limitations |
|---|---|
| • Not limited to experimental doses | • Highly dependent on dose selection |
| • Less dependent on dose spacing | • Highly dependent on sample size |
| • Appropriately accounts for variability and uncertainty resulting from study quality | • Does not account for variability and uncertainty in the experimental results (e.g., does not account for study quality appropriately) |
| • Takes into account the shape of the dose–response curve and other related information | • Dose–response information (e.g., shape of dose–response curve) not taken into account |
| • Corresponds to consistent response level and can be used to compare results across chemicals and studies | • Does not correspond to consistent response levels for comparisons across studies |
| • Flexibility in determining biologically significant rates | • A LOAEL cannot be used to derive a NOAEL |
| BMD limitations | NOAEL advantages |
| • Ability to estimate BMD may be limited by the format of data presented | • Can be used when data is not amenable for BMD modeling |
| • Time consuming | • Easy to derive |
| • More complicated decision making process | • Has been the standard method for deriving a POD for decades (e.g., is familiar to most risk assessors) |
How to Calculate BMDL
We can calculate BMD or BMDL using US EPA's benchmark dose software (BMDS) or RIVM’s PROAST package. Both software packages are internationally recognized. Personally I prefer EPA's software since it is easier to use.
Before we take the BMD approach, we must evaluate whether available toxicology data is suitable for BMD modelling. Available data must meet the following criteria for BMD modelling.
- Reported response data must be either quantal data or continuous data.
- There is a clear dose-response trend.
- There are sufficient dose groups (minimum 3 dosing groups + 1 control group). Datasets in which response is only observed at the high dose are usually not suitable for BMD modeling
- The dose response-model should fit the data adequately by some predefined criterion (e.g. P-value > 0.1).
Once we have determined whether reported response data is quantal or continuous, we can choose dichotomous or continuous models to fit the dose response curve in BMDS. The BMDS software will then perform the fitting and calculation (see picture below).
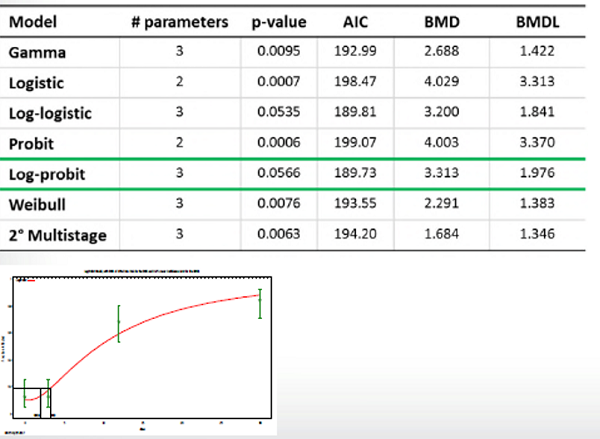
Often, more than one model or modeling options will result in an acceptable fit to the data, current EPA guidance (2012 TG) provides guidance on how to pick a single “best” model:
- What is “sufficiently close” can vary based on the needs of the assessment, but generally should not be more than 3-fold.
- If BMDLs are not sufficiently close, EPA recommends picking the model with the lowest BMDL
- If BMDLs are sufficiently close, EPA recommends selecting the model with the lowest AIC
- If multiple models have the same AIC, EPA recommends combining BMDLs
BMDL vs NOAEL
The value of calculated BMDL can be bigger or smaller than NOAEL. When test sample (i.e, no. of tested animals) is very big, BMDL is bigger than NOAEL. When the size of sample is small, BMDL may be smaller than NOAEL.
References
- Introduction to benchmark dose methods and U.S. EPA's benchmark dose software (BMDS)
- Workshop confirms BMD approach as the best method for dose-response modelling in risk assessment - EFSA
Having Questions?
We do not provide consultancy services. If you have questions or need any help, please contact our sponsor. You may also find an expert in CSP business directory below. If you are a consultant, you may get yourself listed in CSP business directory (free) or sponsor this page to leave your contact info on this page..
