GHS Classification Criteria for Skin Sensitization
Little Pro on 2017-11-08
A chemical substance will be classified as a skin sensitizer if there is reliable human evidence or positive results from an appropriate animal test. In this article, we will compare 3 main skin sensitization test methods, Guinea Pig Maximisation test (GPMT), Buehler test and local lymph node assay (LLNA), and explain what positive result is and how to use the test results (i.e, positive response rate, stimulation index, EC3 value) to conduct GHS classification for skin sensitization.
Comparison of 3 Skin Sensitization Methods
| Test Method & Species | What Is Measured? | What Is Positive? |
|---|---|---|
| Guinea pig maximisation test (GPMT) (OECD 406) | Challenge-induced dermal hypersensitivity reactions elicited in test animals compared with control groups. | In GPMT, Freunds Complete Adjuvant (FCA) is usually injected to potentiate sensitization. A response rate of at least 30% of the animals is considered as positive. |
| Buehler test (OECD 406) - Guinea pig | Challenge-induced dermal hypersensitivity reactions elicited in test animals compared with control groups. | Preferred Buehler test usually does not use adjuvant. A response rate of at least 15% of the animals is considered as positive. |
| Local Lymph Node Assay (LLNA) (OECD 429) - Mice | Lymphocyte proliferation induced in regional lymph nodes in mice. Stimulation Index (SI) at multiple doses: the ratio of the mean proliferation in test group to the mean proliferation in control group. |
Stimulation Index (SI)>=3 is considered positive. EC3 value can be estimated to reflect sensitisation potency. It is the estimated concentration of test chemical required to induce a stimulation index of 3 in the LLNA. |
Note: Due to clear animal welfare benefits and scientific advantages compared with the guinea pig tests, the LLNA is the preferred skin sensitization test method in EU. For example, EC3 value from LLNA test can be used for quantitative health risk assessment.
GHS Classification Criteria for Skin Sensitization
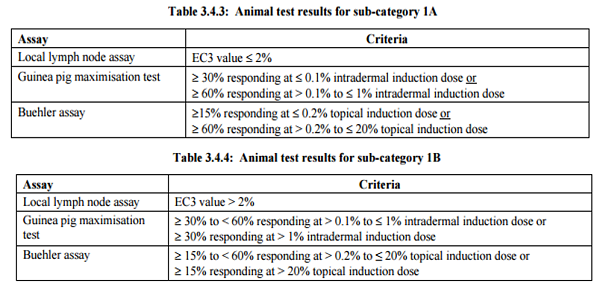
The table below summarizes GHS classification criteria for skin sensitization. There is only 1 hazard category for skin sensitization under GHS. However, data on sensitization potency such as EC3 value and the rate of positive response can be used to divide a sensitizer into sub-category 1A and sub-category 1B.

To be more specific, the table below shows you how to use potency data to sub-categorize a skin sensitizer. It should be noted that only positive response give by intradermal induction in GMPT is considered.
GHS Classification Criteria for Mixture
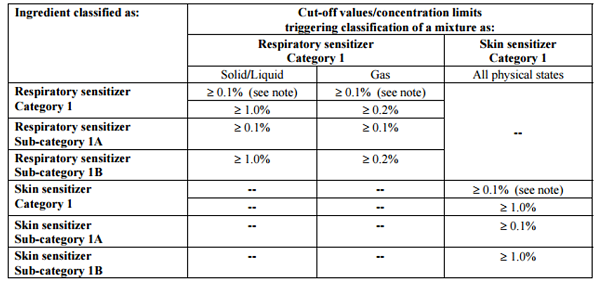
Note: A skin sensitizer generally requires labelling when it is present in a mixture with a concentration above 0.1%.
Reference
- GHS classification criteria - health hazards - UN GHS purple book
- OECD Guidelines 406
- OECD Guidelines 429 - Skin Sensitization
More Readings
- GHS classification criteria in a single page
- UN GHS purple book - health hazards
- GHS classification criteria - acute toxicity
- GHS classification criteria - skin irritation and corrosion
- GHS classification criteria - eye irritation and corrosion
- GHS classification criteria - skin sensitization
- GHS classification criteria - specific target organ toxicity (STOT)
- GHS classification criteria - germ cell mutagenicity
- GHS classification criteria - reproductive toxicity
- GHS classification criteria - carcinogenicity
- M-factor and GHS Classification Criteria for Aquatic Environment
Having Questions?
We do not provide consultancy services. If you have questions or need any help, please contact our sponsor. You may also find an expert in CSP business directory below. If you are a consultant, you may get yourself listed in CSP business directory (free) or sponsor this page to leave your contact info on this page..

Tags: Topics - GHS, GHS Classification