GHS Classification Criteria for Germ Cell Mutagenicity
Little Pro on 2018-03-07
Mutagenicity refers to the induction of permanent transmissible changes in the amount or structure of the genetic material (i.e, gene, DNA) of cells or organisms. A germ cell mutagen is a chemical that may cause mutations in the germ cells of humans that can be transmitted to the progeny.
Genotoxicity is a broader term and refers to processes which alter the structure, information content or segregation of DNA and are not necessarily associated with mutagenicity. All mutagens are genotoxic. Not all genotoxic substances are mutagenic.
Mutations can either occur in germ cells or somatic cells. Under GHS, only germ cell mutagens are classified. However, both test results from in vitro/in vivo germ cell and somatic cell mutagenicity tests should be considered.
- Germ cells are those cells that are involved in the reproductive process and can give rise to a new organism. Male germ cells give rise to sperm and female germ cells develop into ova. Toxicity to germ cells can cause effects on the developing fetus (such as birth defects, abortions).
- Somatic cells are all body cells except the reproductive germ cells. They have two sets (or pairs) of chromosomes. Toxicity to somatic cells causes a variety of toxic effects to the exposed individual (such as dermatitis, death, and cancer).
- In vitro (latin: "in the glass") test is a study that is performed outside of a living organism (i.e, glass, petri dishes). The test usually involves the use of isolated tissues, organs or cells.
- In vivo(latin: "in the living") test is a study that is performed in living organisms (i.e, rat, rabbit).
Hazard Categories for Germ Cell Mutagenicity and GHS Classification Criteria
There are 3 hazard categories for germ cell mutagenicity depending on whether there is sufficient human evidence or animal test data. The table below summarizes GHS classification criteria for tested substances.
| Category | GHS Classification Criteria |
|---|---|
| Category 1A | Chemicals known to induce or regarded as if they induce heritable mutations in human germ cells
Known to induce heritable mutations –positive evidence from human epidemiological studies. |
| Category 1B | Chemicals known to induce or regarded as if they induce heritable mutations in human germ cells
|
| Category 2 | Chemicals that may induce heritable mutations in human germ cells
Positive evidence obtained from experiments in mammals and/or in some cases from in vitro experiments, obtained from:
Note: Chemicals which are positive in in vitro mammalian mutagenicity assays, and which also show chemical structure activity relationship to known germ cell mutagens should be classified as category 2. |
List of Validated Mutagenicity/genotoxicity Tests
The table below summarizes a list of mutagenicity tests and genotoxicity tests that could be used for GHS classification. They are grouped by somatic cells/germ cells and in vitro/in vivo.
| Test Type | Validated Methods |
|---|---|
| Germ cell mutagenicity tests in vivo |
|
| Germ cell genotoxicity tests in vivo |
|
| Somatic cell mutagenicity tests in vivo |
|
| Somatic cell genotoxicity tests in vivo |
|
| In vitro mutagenicity tests |
|
Note:The classification of individual substances should be based on the total weight of evidence available, using expert judgement.
All known germ cell mutagens are also mutagenic in somatic cells in vivo. Substances that are mutagenic in somatic cells may produce heritable effects if they, or their active metabolites, have the capability of interacting with the genetic material of germ cells. Substances that do not induce mutations in somatic cells in vivo would not be expected to be germ cell mutagens.
Examples of Germ Cell Mutagen Classification
| Test Result | Proposed Classification |
|---|---|
| Positive result in a human epidemiological study |
|
| 1 positive result in germ cell mutagencity study in vivo |
|
| 1 positive result in somatic cell mutagencity study in vivo + evidence of exposure of the substance showing activity or metabolite to germ cells |
|
| 1 positive result in somatic cell mutagencity study in vivo |
|
| 1 positive result in somatic cell genotoxicity study in vivo + 1 positive result in in vitro mutagenicity study |
|
| Only negative results in in vivo mutagenicity tests |
|
| Only 1 postive result in in vitro mutagenicity test data available |
|
| Negative result in the in vivo genotoxicity test. However, in vivo mutagenicity data is not available. |
|
Mutagen Classification for Mixture
For a mixture containing mutagens, the mixtures may be classified using the table below:
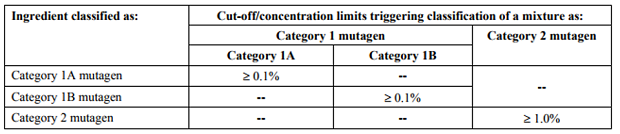
Read more: comparison fo GHS concentration limits for mutagenicity
References and More Reading
- GHS classification criteria in a single page
- UN GHS purple book - health hazards
- GHS classification criteria - acute toxicity
- GHS classification criteria - skin irritation and corrosion
- GHS classification criteria - eye irritation and corrosion
- GHS classification criteria - skin sensitization
- GHS classification criteria - specific target organ toxicity (STOT)
- GHS classification criteria - germ cell mutagenicity
- GHS classification criteria - reproductive toxicity
- GHS classification criteria - carcinogenicity
- M-factor and GHS Classification Criteria for Aquatic Environment
Having Questions?
We do not provide consultancy services. If you have questions or need any help, please contact our sponsor. You may also find an expert in CSP business directory below. If you are a consultant, you may get yourself listed in CSP business directory (free) or sponsor this page to leave your contact info on this page..

Tags: Topics - GHS, GHS Classification