Acute Toxicity
Little Pro on 2016-04-24
Acute toxicity describes the adverse effects of a substance that result either from a single exposure or from multiple exposures in a short period of time (usually less than 24 hours). Acute toxicity tests in animals (i.e, rat) use mortality as the main observational endpoint in order to derive a LD50 or LC50.
Acute toxicitity studies usually include 3 routes: oral, dermal and inhalation.
Dose Descriptor for Acute Toxicity
A common dose-response descriptor for acute toxicity is the LD50 (Lethal Dose 50%). This is a statistically derived dose at which 50% of the individuals will be expected to die. For inhalation toxicity, air concentrations are used for exposure values. Thus, the LC50 (Lethal Concentration 50%) is used.
The units of LD50 and LC50 are listed as follows:
- LD50: mg/kg/bw. mg/kg bw/d stands for mg of substance per kg of body weight administered per day.
- LC50:mg/L. mg/L is the estimated air concentration of a substance administered via inhalation route.
It shall be noted that LD50/LC50 from acute toxicity studies are mainly used for GHS acute toxicity classification, qualitative risk assessment and dose selection for repeated dose toxicity studies. They cannot be used to derive No-observed-adverse-effect-level (NOAEL).
GHS Classification Criteria for Acute Toxicity
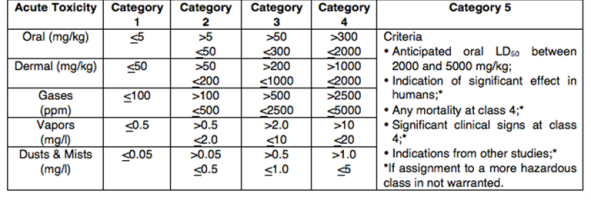
LD50 and LC50 values are used to describe the relative acute toxicity of a chemical substance. The table below shows GHS classification criteria for acute toxicity. Acute toxicity category 1 represents the most severe toxicity.
Acute Toxicity Testing Guidelines
- OECD TG 402 Acute Dermal Toxicity
- OECD TG 403 Acute Inhalation Toxicity
- OECD TG 420 Acute Oral Toxicity-FDP
- OECD TG 423 Acute Oral Toxicity-ATC 425
- OECD TG Acute Oral Toxicity: Up-and-Down
- OECD TG 436 Acute Inhalation Toxicity - ATC
 |
Congratulations! You have learned the definition of acute toxicity, dose descriptor for acute toxicity (LD50 and LC50), GHS classification criteria for acute toxicity and testing guidelines. |
Continue Learning...
- Acute toxicity (oral, inhalation, dermal)
- Skin and eye irritation
- Sensitization (skin, respiratory)
- Repeated dose toxicity (28d, 90d and chronic)
- Mutagenicity and genotoxicity
- Reproductive and developmental toxicity
- Carcinogenicity
- Toxicokinetics
Having Questions?
We do not provide consultancy services. If you have questions or need any help, please contact our sponsor. You may also find an expert in CSP business directory below. If you are a consultant, you may get yourself listed in CSP business directory (free) or sponsor this page to leave your contact info on this page..
