Positive List of Food Contact Substances in EU
Little Pro on 2017-02-14
In EU, Regulation (EU) No 10/2011 on plastic materials and articles (plastics regulation) sets out rules on the composition of plastic food contact materials (FCMs), and establishes a Union List of substances that are permitted for use in the manufacture of plastic FCMs. Regulation (EC) No 450/2009 foresees the establishment of a Union list of substances permitted for the manufacture of active and intelligent materials. However, the list is not published yet. For other groups of food contact materials (i.e, adhesives), there is no positive list of food contact substances at the EU level. However, national authorities may have set their own lists.
Positive List of Food Contact Substances for Plastics in EU
The annex I to the plastics regulation is the Union list of authorised substances that can be used in the manufacture of the plastic layers of food contact plastic materials and articles (see picture below). The list covers monomers, starting substances, additives and polymer processing aids.
You have two options to search the list:
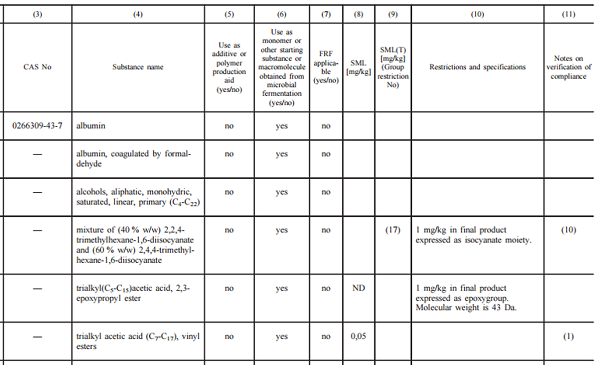
Note 1: This positive list does not include authorized solvents and colorants, and substances only used to manufacture adhesives, coatings and printing inks. Substances on provisional list may also be used if compliant with national legislation.
Note 2: In the Union list, only the name of the free acid, alcohol or phenol is mentioned. However, the use of certain salts of these acids, alcohols or phenols is also authorised. The salts of the following cations can be used without any restriction: aluminium, ammonium, calcium, magnesium, potassium, and sodium.
Column 8 & 9: If a listed substance is used in the final plastic food contact material, it has to comply with the specific migration limit (SML) for this substance listed in column 8 and the group restriction listed in column 9 (applicable to a group of substances). For substances for which no SML is established, the specific migration of these substances shall not exceed a generic SML of 60 mg/kg.
Column 10: A listed substance must also comply with restriction and specification set out in the column 10 (i.e, "not to be used for articles in contact with fatty foods").
Column 11: Attention shall also be paid to the notes on verification of compliance given in the column 11 (see example below).
| Note No. | Notes on verification of compliance |
|---|---|
| 1 | Verification of compliance by residual content per food contact surface area (QMA) pending the availability of an analytical method. |
| 2 | There is a risk that the SML or OML could be exceeded in fatty food simulants. |
| 3 | There is a risk that the migration of the substance deteriorates the organoleptic characteristics of the food in contact and then, that the final product does not comply with Article 3(1) c of the Framework Regulation (EC) No 1935/2004. |
| 4 | Compliance testing when there is a fat contact should be performed using saturated fatty food simulants as simulant D2. |
| 5 | Compliance testing when there is a fat contact should be performed using isooctane as substitute of simulant D2 (unstable). |
| 6 | Migration limit might be exceeded at very high temperature. |
New Food Contact Substance Notification in EU
New substances to be used to manufacture plastic food contact materials can be added to the positive list by following the procedure below.
- Applicant submit an application to the competent authority of a Member State by providing a technical dossier containing the information specified in the guidelines for the safety assessment of a food contact additive and a summary of the technical dossier.
- The national contact points will then forward the application to the European Food Safety Authority (EFSA). EFSA will check the validity of the application according to the EFSA Guidance and make proposals.
Positive List for Other Food Contact Materials
For some types of food contact materials (i.e, coatings, adhesives, and paper) for which there is no Union list of authorised substances, some EU Member States have set their own positive lists. More info can be found below.
Reference
How to Comply with Food Contact Regulations in EU
Having Questions?
We do not provide consultancy services. If you have questions or need any help, please contact our sponsor. You may also find an expert in CSP business directory below. If you are a consultant, you may get yourself listed in CSP business directory (free) or sponsor this page to leave your contact info on this page..

Tags: Topics - Food Contact, Food Contact Regulations