EU REACH Registration 2018
Little Pro on 2015-12-30
REACH registration is required for all substances manufactured in EU or imported into EU in quantities of one tonne or more per year. This includes substances on their own, in mixtures or substances in articles when they are intended to be released under normal conditions of use. The last REACH registration deadline is 31 May 2018 for all >=1t/y substances.
For all registrations, a registration dossier has to be prepared and submitted electronically via REACH-IT to the European Chemicals Agency (ECHA). Normally, the registration must be successfully completed and a REACH registration number assigned to the registrant before a substance can be manufactured, imported or placed on the market.
Which substances need to be registered under REACH?
Registration is required for all substances manufactured or imported in quantities of one tonne or more per year per manufacturer or importer unless they are exempted. For polymers, monomers shall be registered.
The following substances are exempt from REACH registration:
- Substances exempted from REACH regulation: radio-active substances, substances under customs supervision, substances used for national defense, non-isolated intermediates, wastes, etc.;
- Substances regulated by other legislation: substances in food, medicinal products, plant protection products, etc.;
- Substances in REACH annex VI: a list of exempt substances well-known to be safe (i.e., water, nitrogen);
- Substances covered by REACH annex V: by-products, naturally occurring substances, impurities, etc.;
Who needs to register?
The following actors in the supply chain must comply with REACH registration requirements:
- EU manufacturers and importers of substances on their own or in a mixture; or
- EU producers and importers of articles containing substances intended to be released; or
- "Only representatives" established in the EU and appointed by a manufacturer, formulator or article producer established outside the EU;
Only Representative: Under REACH, a natural or legal person established outside the EU, who manufactures a substance, formulates a mixture or produces an article can appoint an only representative to carry out the required registration of the substance that is imported (as such, in a mixture or in an article) into the EU (Article 8(1)). This will relieve the EU importers within the same supply chain from their registration obligations, as they will be regarded as downstream users.
REACH registration deadline
Different deadlines have been set for phase-in substances and non-phase-in substances under REACH. Phase-in substances have to be registered according to the following schedule if they were pre-registered before pre-registration deadline 1 Dec 2008 while non-phase-in substances need to be registered immediately. The last registration deadline is 31 May 2018 for all pre-registered phase-in substances.
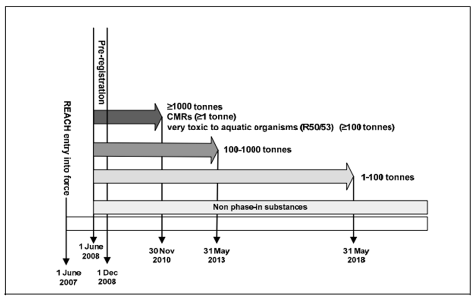
Phase-in substances are mainly substances already manufactured or placed on the market before REACH's entry into force. Substances fulfilling at least one of the following criteria may be considered as phase-in substances.
- Substances listed in the European Inventory of Existing Commercial Chemical Substances (EINECS);
- Substances listed in the European List of Notified Chemical Substances (ELINCS);
- Substances listed in the No-longer Polymer List (NPL);
Non-phase-in substances are all other substances that do not fulfil any of the criteria above and they cannot enjoy extended registration deadlines. Potential manufacturers and importers of non-phase-in substances have to submit an inquiry to ECHA and subsequently register the substance first before they can manufacture or import the substance.
What if I have missed pre-registration deadline?
Phase-in substances that missed pre-registration deadline (1 Dec 2008) cannot benefit from extended registration deadlines and need to be registered without any delay.
Potential registrants who, for the first time after 1 December 2008, manufacture or import a phase-in substance in quantities of one tonne per year or more can still submit certain information to ECHA (late pre-registration) and benefit from the extended deadlines. Late pre-registrations have to be submitted within six months after the manufacturing or importing of the substance that exceeds the one-tonne threshold and no later than twelve months before the relevant registration deadline.
Type of Registrations under EU REACH
| Pre-registration |
|
| Late Pre-registration |
|
| Registration |
|
| Inquiry |
|
| Intermediate Registration |
|
| PPORD Notification |
|
REACH Annex VII has set standard information requirement on REACH registration. This annex VII can be downloaded from here.
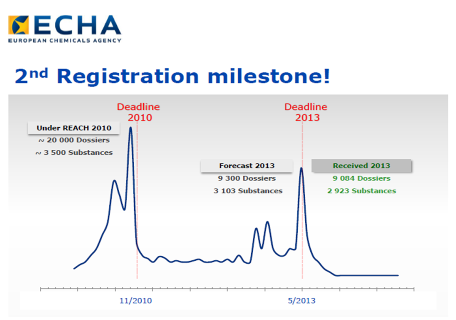
Statistics on REACH Registrations
Click the picture below to find out how many registrations have been submitted and whether your substance has been registered under EU REACH or not.
Reference & Resources
Click here to access all references and resources for Europe including the English translation of regulations, regulatory lists and useful links to the websites of competent authorities.
Having Questions?
We do not provide consultancy services. If you have questions or need any help, please contact our sponsor. You may also find an expert in CSP business directory below. If you are a consultant, you may get yourself listed in CSP business directory (free) or sponsor this page to leave your contact info on this page..