How to Modify Dose Descriptors to A Correct Starting Point
Little Pro on 2016-08-10
When calculating derived no-effect level (DNEL), it is very important to convert certain dose descriptors (e.g. NOAEL, benchmark dose, LD/LC50) into a correct starting point. The goal is to get the corrected N(L)OAEL which matches real human exposure conditions and which is in the right unit. This process is called modification of dose descriptors. DNELs will be calculated by dividing corrected dose descriptors with appropriate assessment factors. In this article, we will show you why the modification of dose descriptors is necessary and how to modify dose descriptors. Examples and quiz will be given.
When is modification of dose descriptors necessary?
Modification of dose descriptor to a correct starting point is necessary if there are any differences below between exposure routes and between experimental animals and humans.
- Differences in bioavailability (e.g., differences in absorption between routes and between experimental animals and humans).
- Differences in the units of exposures in case of route-to-route extrapolation (e.g, converting on oral NOAEL in mg/kg bw to inhalatory NOAEC in mg/m3).
- Differences in human and experimental exposure conditions (e.g. 6h/d for rat and 8h/d for workers).
- Differences in respiratory volumes between experimental animals (at rest) and humans (light activity).
It should be noted that modification is not appropriate in cases where the dose descriptor is based on human data or where human exposure is evaluated based on biological monitoring data.
Example 1 Converting an oral/dermal NOAEL rat to a corrected oral/dermal NOAEL rat
When there is a difference in absorption between routes or between experimental animals and humans, modification of starting point is necessary. The picture below shows how to get a corrected NOAEL for oral or dermal route from available NOAEL data. The unit of both oral and dermal NOAEL is mg/kg body weight/day.

Note 1: In the absence of absorption data for both the starting route and the target route, worst case assumptions have to be made. Worst case will be obtained assuming a limited absorption for the starting route and full absorption for the target route (100%), leading to a low (conservative) NOAEL. If there is reliable toxico-kinetics data, the actual absorption rate can be used.
For example, if a rat oral NOAEL is 100mg/kg/bw and toxicokinetics data shows that the oral absorption in rat is only 20%, the corrected NOAEL will be 20mg/kg/bw (assuming 100% absorption for human).
Note 2: Above equation only works for oral and dermal route. For inhalation route, please see example 2 and 3.
Example 2 Converting an inhalatory NOAEC rat to a corrected inhalatory NOAEC
Inhalatory NOAEC rat (in mg/m3) usually need to be corrected for differences in inhalation absorption between rats and humans, the differences in exposure conditions (6h/d for rat, 8h/d for workers and 24h/d for general population). For workers, the differences between respiratory rates under standard conditions and under conditions of light activity also need to be considered.
The picture below shows you how to modify inhalatory NOAEL to a correct starting point when deriving DNELs for workers and general population for inhalation route.
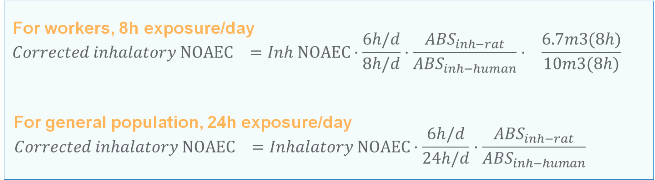
Note 1: Default inhalation absorption rate for both humans and rat is 100%. If there is reliable toxico-kinetics data, the actual absorption rate can be used.
Example: If NOAEC (rat, inhalation route) was 80mg/m3, then the corrected NOAEC for deriving DNEL for workers for inhalation route would be 40.2mg/m3 assuming 100% absorption for both rat and humans. The corrected NOAEC for deriving DNELs for general population would be 20mg/m3.
Example 3 Converting an oral NOAEL rat (in mg/kg bw/day) to a corrected inhalatory NOAEC (in mg/m3)
When converting oral N(L)OAEL rat (in mg/kg bw/day) into inhalatory N(L)OAEC rat (in mg/m3), one must take into account of all differences in absorption rate, exposure conditions, respiratory rate and unit. The picture below shows you how to calculate corrected NOAEC (in mg/m3) from oral NOAEL rat when deriving DNELs for workers and general population for inhalation route.
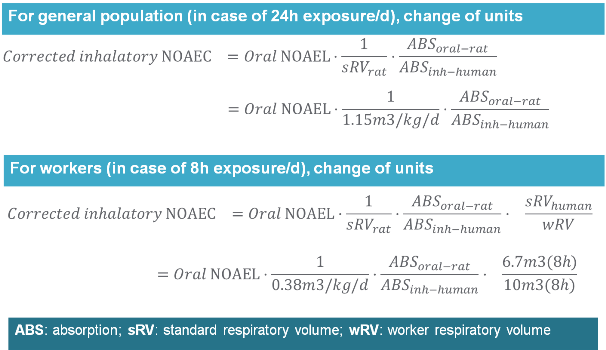
Example: If NOAEL (rat, oral route) was 115mg/kg bw/day, then the corrected NOAEC for deriving DNEL for workers for inhalation route would be 101.3mg/m3 assuming 50% oral absorption and 100% inhalation absorption (instead of 100% absorption for oral route). The corrected NOAEC for deriving DNELs for general population for inhalation route would be 50mg/m3.
Note 1: Converting dermal NOAEL to corrected NOAEC is similar. If you reverse above equation, you can also calculate oral or dermal NOAEL from NOAEC values.
Note 2: Allometric scaling factors (interspecies AF=4) do not need to be applied to the corrected NOAEC (in mg/m3) when deriving DNELs for inhalation route.
Example 4 Converting dermal NOAEL (in mg/kg bw/day) to dermal NOAEL (in mg/cm2/day)
The dose descriptor for dermal irritation/corrosion may come from dermal sub-acute or sub-chronic studies. In these studies, the dose descriptor is usually reported in mg/kg body weight/day. This needs to be modified to enable comparison with the human exposure, generally expressed in mg/cm2/day. The equation below shows you how to convert dermal NOAEL to a different unit.
Corrected NOAEL=NOAEL*body weight (in kg)/total application area(in cm2)
Let's assume that:
- the average weight of rats is 0.25kg;
- the dose is applied to approximately 10% of the total body surface, and
- the total body surface of rats is on the average 445 cm2.
If the highest dose not causing irritation/corrosion to rat was 100 mg/kg bw in the sub-acute study, the modified dose descriptor would be 100 mg/kg*0.25kg/44.5 cm2 = 0.56mg/cm2 = 560μg/cm2.
Quiz
Q1: Is modification of dose descriptor necessary if using oral NOAEL (rat) to derive DNELs for oral route for general population?
- Yes.
- No.
- It depends. If there is no difference in oral absorption rate between rats and humans, there is no need to modify dose descriptors.
Q2: Is modification of dose descriptor necessary if using inhalatory NOAEL (rat) to derive DNELs for inhalation route for workers and general population?
- Yes.
- No.
- It depends. If there is no difference in absorption rate between rats and humans, there is no need to modify dose descriptors.
Q3: What is corrected dose descriptor (NOAEL) for deriving DNELs for oral route for humans based on the the following data?
- Dermal NOAEL(rat) from a repeated dose toxicity study =50mg/kg bw/day.
- Dermal absorption rate for rat from toxicokinetics study=10%. .
View quiz answer and explanation
Main Reference

Good job. You have learned why the modification of dose descriptors is important and how to modify dose descriptors to correct starting point. Please subscribe our newsletter to keep updated of our new articles.
"It does not matter how slowly you go as long as you do not stop. "
– Confucius
Having Questions?
We do not provide consultancy services. If you have questions or need any help, please contact our sponsor. You may also find an expert in CSP business directory below. If you are a consultant, you may get yourself listed in CSP business directory (free) or sponsor this page to leave your contact info on this page..
