US Toxic Substances Control Act (TSCA)
Little Pro on 2015-12-30
The Toxic Substance Control Act (TSCA) is the most important chemical control law in the United States. It has given Environmental Protection Administration (EPA) various authorities to take certain regulatory actions against both new and existing chemical substances. In 2016, TSCA was amended by "The Frank R. Lautenberg Chemical Safety for the 21st Century Act". In this article, we will summarize the main provisions of TSCA, pre-manufacture notice (PMN), significant new use rules (SNUR), and main changes in revised TSCA.
Note: As an "industrial chemical substance" law, TSCA does not apply to certain tobacco products, nuclear materials, munitions, foods, food additives, drugs, cosmetics, and substances used solely as pesticides.
Main Provisions of TSCA
| Section | Requirement and Notes |
|---|---|
| Section 4 Testing for Existing Chemicals |
|
| Section 5 New Chemicals Review |
|
| Section 5 Significant New Use Rules (SNUR) |
|
| Section 6 Regulation of Hazardous Substances |
|
| Section 8 Reporting and Record-keeping |
|
| Section 13 Import |
|
Pre-manufacture Notice (PMN) for New Substances
Section 5 of TSCA requires anyone who plans to manufacture or import a new chemical substance to obtain EPA's approval by submitting a pre-manufacture notice (PMN) at least 90 days prior to the manufacture or import. A new substance is defined as one substance that is not listed in TSCA Inventory of Chemical Substances.
Applicant must submit completed PMN to EPA electronically with available data and wait for EPA's 90 day review. Toxicology data is not mandatory, but must be submitted if available.
The possible outcomes of EPA's PMN review include:
- Drop or approval: no further regulatory actions (about 90% of PMNs belong to this case);
- Withdrawal;
- Request additional data;
- Approval with some type of restrictions (Consent orders);
- Approval with Significant New Use Rules (SNURs) establishing some type of restrictions. The restrictions apply to both the submitter and other manufacturers and importers of the same substance.
PMN Exclusions and Exemptions
The following substances are excluded or fully exempt from PMN reporting:
- naturally-occurring materials;
- products of incidental reactions;
- products of end-use reactions;
- mixtures (but not mixture components);
- impurities or byproducts;
- substances manufactured solely for export;
- non-isolated intermediates;
- substances formed during the manufacture of an article.
There are other types of PMN exemptions while some exemptions require EPA's approval (i.e., LVE, LoREX and TME). The table below summarizes the special types of PMN exemptions.
| Research & Development Exemption |
|
| Polymer Exemption |
|
| Low Volume Exemption (LVE) |
|
| Low Releases and Low Exposures (LoREX) |
|
| Test Marketing Exemption (TME) |
|
PMN and Notice of Commencement (NOC)
When a PMN is approved, a 'Notice of Commencement' (NOC) is required to be submitted to EPA within 30 days of first importation or production. Following receipt of this NOC, the subject chemical substance will be added to EPA's TSCA Inventory of existing chemical substances for the purposes of future importation and/or domestic production.
It shall be noted that substances that benefit from PMN exemptions do not require NOC and will not be added onto TSCA inventory. However, TSCA certification is stilled required for importing those chemical substances to the United States.
TSCA Section 5 (e) Consent Orders
Consent Orders are PMN approvals with restrictions. For example, the orders may require the PMN submitter to take certain risk management measures to minimize exposure and risks. A chemical substance must be in compliance with the provisions of the consent order before it can be manufactured or imported. TSCA section 5(e) Consent Orders are only binding on the original PMN submitter.
Significant New Use Rules (SNURs)
Section 5 of TSCA authorizes EPA to designate use of a chemical substance as a 'significant new use', and require the submission of information to EPA prior to the chemical substance being manufactured (including imported) or processed for that use. Unlike consent order which is only binding on the PMN submitter, SNURs apply to all manufactures and importers of the same substance.
Substances that are subject to SNUR requirements are designated as such by an 'S' flag in the TSCA Inventory. If your chemical substance is subject to a SNUR and your intended manufacture, processing, or use of the substance is a significant new use, you would be required to submit a Significant New Use Notice (SNUN) 90 days prior to the manufacture of that substance.
SNUNs are reported using the standard PMN form and are subject to a 90-day review process similar to that for a PMN. When submitting a SNUN, the submitter should include a cover letter that provides the CFR citation of the SNUR and identifies the specific significant new use(s) for which the SNUN is being submitted.
Chemical Data Reporting (CDR)
The Chemical Data Reporting (CDR) Rules, previous known as Inventory Updating Reporting (IUR) Rules, require manufacturers and importers to report if, for any calendar year since 2011, a chemical substance was manufactured (including imported) at a site in production volumes of 25,000 lbs. CDR is conducted every 5 years.
TSCA Inventory Reset Rule
Amended Toxic Substances Control Act (TSCA) requires EPA to designate chemical substances on the TSCA Chemical Substance Inventory as either “active” or “inactive” in U.S. commerce. Substances designated as "active" on TSCA inventory can be manufactured, imported or processed without any notifications. Substances designated as "inactive" on TSCA inventory must be notified to the US EPA via Notice of Activity Form (NOA-B) 90 days prior to planned manufacture, import or processing.
TSCA Certification for Imported Substances
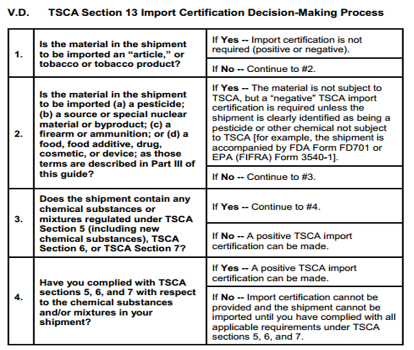
TSCA section 13 requires that importers 'certify' their imported chemical substances or mixtures are either: (1) in compliance with TSCA Sections 5, 6 and 7 at the time of import; or (2) not subject to TSCA. Customs can refuse entry of any shipment that does not have a TSCA Certificate of Compliance. This certificate could be a self-declaration.
Some products do not require TSCA certificate. The picture below shows you whether a TSCA compliance certificate is needed.
Old TSCA 1976 vs New TSCA 2016
| Item | New TSCA 2016 | Old TSCA 1976 |
|---|---|---|
| Existing Chemicals Review | Mandatory duty on EPA to evaluate existing chemicals with clear and enforceable deadlines | No duty to review; no deadlines for action |
| Assessment Standard | Chemicals assessed against a risk-based safety standard | Against risk-balancing standard |
| New Chemicals Review & SNURS | Requires EPA to make an affirmative determination on new chemicals and existing substances subject to SNURs before entry into the marketplace | Chemicals can enter the market in the absence of EPA action |
| TSCA Inventory | Requires industry to report on the chemicals they manufactured or processed in previous 10 years to determine if chemicals are currently “active” in the marketplace. Chemicals on TSCA inventory will be set as "active" or "inactive". | Substances on TSCA inventory are not set as "active" or "inactive" |
Related Regulations and Standards
Reference & Resources
Click here to access all chemical regulatory references and resources for the United States.
Having Questions?
We do not provide consultancy services. If you have questions or need any help, please contact our sponsor. You may also find an expert in CSP business directory below. If you are a consultant, you may get yourself listed in CSP business directory (free) or sponsor this page to leave your contact info on this page..
