CMR Category 1A/1B Substances
Little Pro on 2017-04-11
The entry 28, 29 and 30 of REACH annex XVII restricts the use of CMR category 1A and 1B substances in products supplied to the general public (i.e, cleaning products, paints) and requires additional labeling for products intended for professional users. In this article, we have summarized how to find out if a substance is restricted, what restriction conditions are and if there are any exemptions.
Definition of CMR Category 1A/1B Substances
CMR substances are substances that are carcinogenic, mutagenic or toxic to reproduction (CMR). They are of specific concern due to the long term and serious effects that they may exert on human health. Under GHS, CMR substances can be classified into 3 categories depending on the severity of hazards.
- Category 1A: Known human carcinogen (H340), mutagen (H350) or reproductive toxicant (H360) based on human evidence ;
- Category 1B: Presumed human carcinogen (H340), mutagen (H350) or reproductive toxicant (H360) based on animal studies;
- Category 2: Suspected carcinogen (H341), mutagen (H351) or reproductive toxicant (H361) based on limited evidence from animal studies or/and human.
Not all CMR substances are restricted by REACH. Only CMR category 1A/1B substances listed in the table 3.1 of annex VI to CLP regulation (see table below) are restricted by REACH. You can click here to download the complete table. If any substance has been assigned with hazard statement code H340, H350 or H360, it will be restricted by REACH.
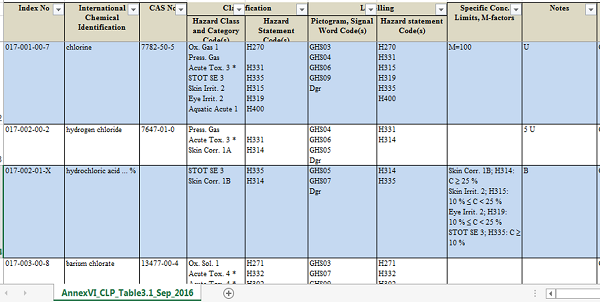
Note:The table 3.1 of Annex VI to CLP is not a static table. Once a substance has received new harmonised classifications as CMR category 1A or 1B based on human and animal evidence respectively, it will be added to the table 3.1.
Conditions of Restriction for CMR 1A/1B Substances in REACH Annex XVII
The table below summarizes the restriction conditions for CMR category 1A/1B substances in REACH Annex XVII.
| Entry | Requirements and Exemptions |
|---|---|
| 28, 29,and 30 for CMR 1A/1B substances listed in the table 3.1 of annex VI of CLP regulation |
1. Shall not be placed on the market, or used, — as substances, — as constituents of other substances, or, — in mixtures, for supply to the general public when the individual concentration in the substance or mixture is equal to or greater than: — either the relevant specific concentration limit specified in Part 3 of Annex VI to Regulation (EC) No 1272/2008, or, — the relevant generic concentration limit specified in Part 3 of Annex I of CLP Regulation (EC) No 1272/2008. [Comment by CSP: The use of a CMR substance in article is excluded from restriction. The CLP generic concentration limit is 0.1% for carcinogen or mutagen and 0.3% for reproductive toxicant.] Without prejudice to the implementation of other Community provisions relating to the classification, packaging and labelling of substances and mixtures, suppliers shall ensure before the placing on the market that the packaging of such substances and mixtures is marked visibly, legibly and indelibly as follows: ‘Restricted to professional users’. 2. By way of derogation, paragraph 1 shall not apply to: (a) medicinal or veterinary products as defined by Directive 2001/82/EC and Directive 2001/83/EC; (b) cosmetic products as defined by Directive 76/768/EEC; (c) the following fuels and oil products: — motor fuels which are covered by Directive 98/70/EC, — mineral oil products intended for use as fuel in mobile or fixed combustion plants, — fuels sold in closed systems (e.g. liquid gas bottles); (d) artists’ paints covered by Regulation (EC) No 1272/2008; (e) the substances listed in Appendix 11, column 1, for the applications or uses listed in Appendix 11, column 2. Where a date is specified in column 2 of Appendix 11, the derogation shall apply until the said date. |
What Is New?
3 April 2017, EU member state officials have voted to unanimously approve the addition of 25 carcinogenic, mutagenic or reprotoxic (CMR) substances to REACH Annex XVII – the restricted substances list. The substances include bisphenol A and ?phenol, (tetrapropenyl) derivatives. Read more..
Having Questions?
We do not provide consultancy services. If you have questions or need any help, please contact our sponsor. You may also find an expert in CSP business directory below. If you are a consultant, you may get yourself listed in CSP business directory (free) or sponsor this page to leave your contact info on this page..

Tags: Topics - Restriction, Substance Restriction in Articles