REACH SVHC List 2023
Little Pro on 2015-12-30
Substances fulfilling one or more of the criteria defined in Article 57 of the EU REACH Regulation can be identified as "substances of very high concern" (SVHC) and put on the "candidate List for authorization" which is also called "REACH SVHC list".
Substances on REACH SVHC list are:
- substances meeting the criteria for classification as carcinogenic, mutagenic or reprotoxic (CMR) category 1 or 2;
- persistent, bio-accumulative and toxic (PBT) substances; or
- very persistent and very bio-accumulative (vPvB) substances;
- substances for which there is evidence for similar concern, such as endocrine disruptors.
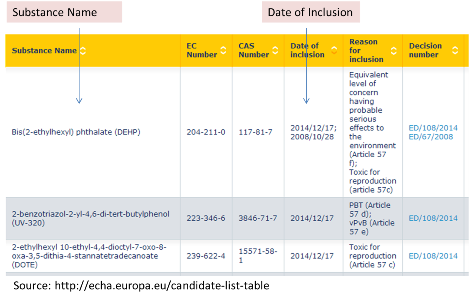
Download the Latest REACH SVHC List in Excel Table
REACH SVHC list is not a static list and it is updated frequently. Up to 17 Jan 2023, there are 233 substances/entries on the SVHC candidate list. Please click the picture below to download the latest REACH SVHC list in excel table. The date of inclusion is also listed.
Note: If a substance is added to SVHC list, that does not mean it is banned. However, if the substance is further added to REACH authorization list, it cannot be placed on the EU market or used after a given date, unless an authorization is granted for their specific use, or the use is exempted from authorisation.
We have launched a free tool called REACH SVHC Finder to help you quickly search if a chemical substance belongs to Substance of Very High Concern (SVHC) under EU REACH regulation or not and whether it is subject to REACH authorization. The tool allows you to search REACH SVHC list and REACH authorization list at the same time.
From SVHC Candidate List to Authorization List
Up to Jan 2023, 59 substances from SVHC candidate list have subsequently been included in the Authorisation List. All substances on the Authorisation list are SVHCs.
What does REACH SVHC list mean to you?
You shall closely monitor the update of SVHC list. You may have immediate legal obligations following inclusion of the substance on the REACH SVHC list.
If your article contains substance(s) included in the SVHC list in concentration above 0.1% (w/w), you need to fulfill the following obligations:
- Duty to communication information on SVHCs - REACH article 33;
- Notification of SVHC in articles to ECHA- REACH article 7(2);
It shall be noted that you also need to check if your products contain any substances on REACH restricted substance list.
Check out a good example of REACH Certificate of Compliance.
Article 33 - duty to communicate information of substances in articles
EU producers or importers of articles which contain substances on the SVHC list in a concentration above 0.1% (w/w) have to provide sufficient information to their customers to allow safe use of the article as soon as the substances are included or, upon request, to a consumer within 45 days of the receipt of the request.
Note that the term "customers" refers to industrial or professional users and distributors, but not to consumers. If no particular information is necessary to allow safe use of the article containing a substance from the SVHC list, as a minimum the name of the substance in question has to be communicated.
REACH does not specify a format for providing information according to Article 33. ECHA's guidance has listed several possible formats:
- Standard answering letters;
- Instructions for use;
- Info on labels;
- Standard communication formats developed by industry associations.
Article 7(2) - notification of substances in articles
EU and EEA producers or importers of articles have to notify ECHA if their article contains a substance on the SVHC list. This obligation applies if the substance is present in those articles in quantities totalling over 1t/y per producer or importer, and if the substance is present in those articles above a concentration of 0.1% (w/w).
- A notification has to be submitted no later than 6 months after its inclusion;
- A notification is not required for a substance in articles which have been produced or imported before the substance has been included on the SVHC list.
- According the latest ruling of the European Court of Justice (ECJ), the 0.1% threshold for notifying SVHCs in articles applies to “each of the articles incorporated as a component of a complex product” rather than to the entire article.
- The obligation to notify substances in articles also applies to packaging materials. Packaging is to be assessed separately from any object it contains.
SVHC Notification Exemptions
- The producer or importer of an article can exclude the exposure of humans and the environment to the substance during normal or reasonably foreseeable conditions of use of the article, including its disposal; or
- The substance has already been registered by a manufacturer or importer in the EU for that use.
What if my article does not contain substances on REACH SVHC list?
You should check whether any of the substance included in your article is subject to restriction under REACH. If yes, you need to comply with restriction conditions set in REACH annex XVII.
Check out a good example of REACH Certificate of Compliance.
Reference & Resources
Click here to access all references and resources for Europe including the English translation of regulations, regulatory lists and useful links to the websites of competent authorities.
Having Questions?
We do not provide consultancy services. If you have questions or need any help, please contact our sponsor. You may also find an expert in CSP business directory below. If you are a consultant, you may get yourself listed in CSP business directory (free) or sponsor this page to leave your contact info on this page..

Tags: Topics - Restriction, Substance Restriction in Articles